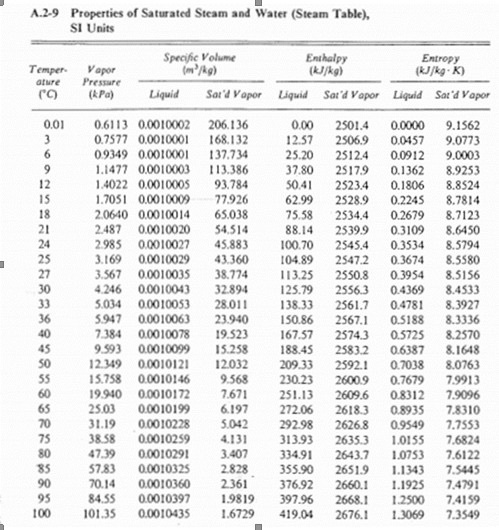
SOLVED: Sodium hydride reacts with excess water to produce aqueous sodium hydroxide and hydrogen gas. How many grams of NaH will produce 0.982 L of gas at 301 K and barometric pressure
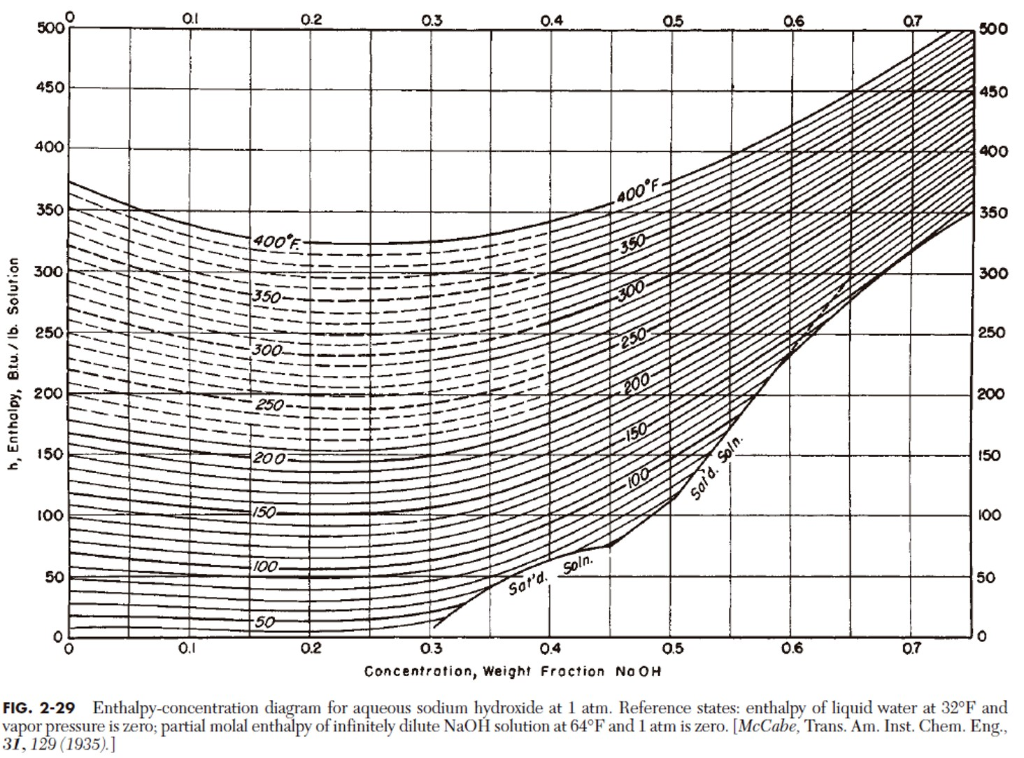
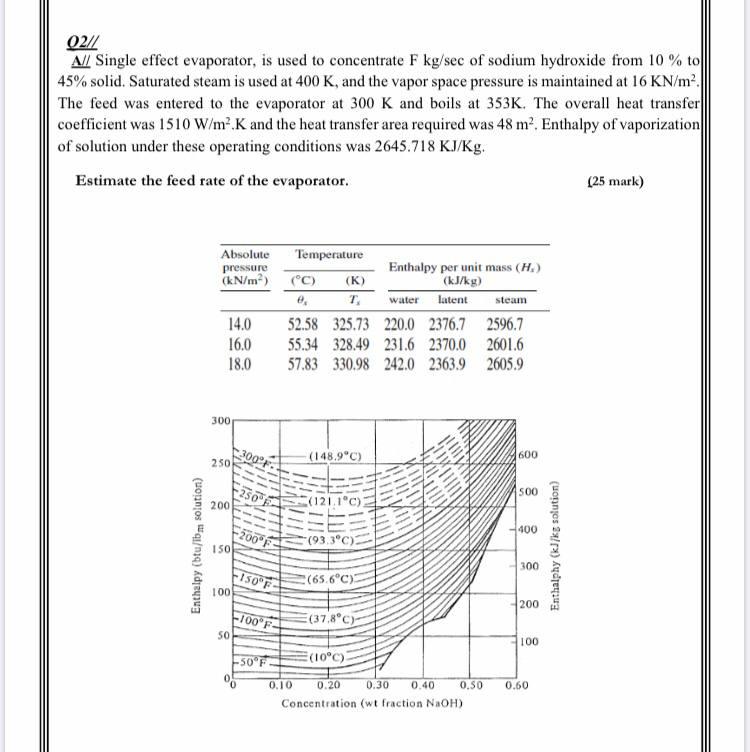
Title P-V-T relations of aqueous sodium hydroxide and its causticity at high temperatures and high pressures Author(s) Kiyama, R
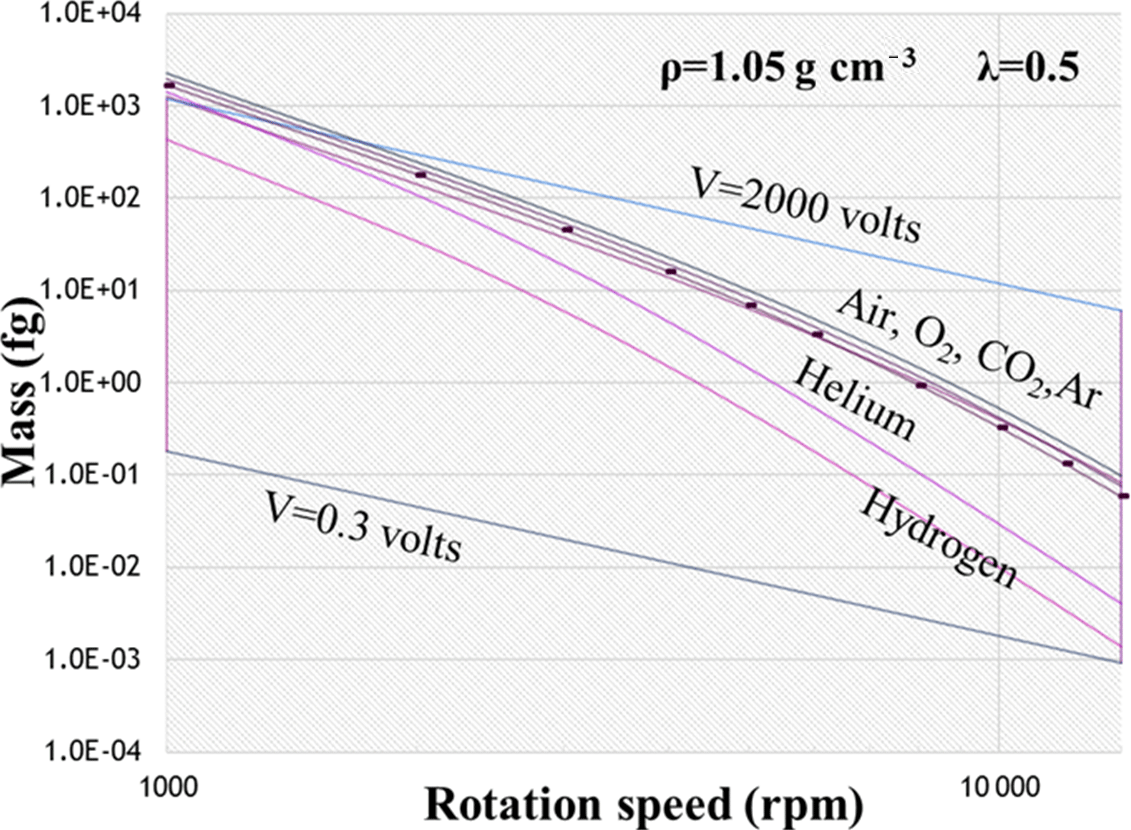
AMT - Effects of temperature, pressure, and carrier gases on the performance of an aerosol particle mass analyser
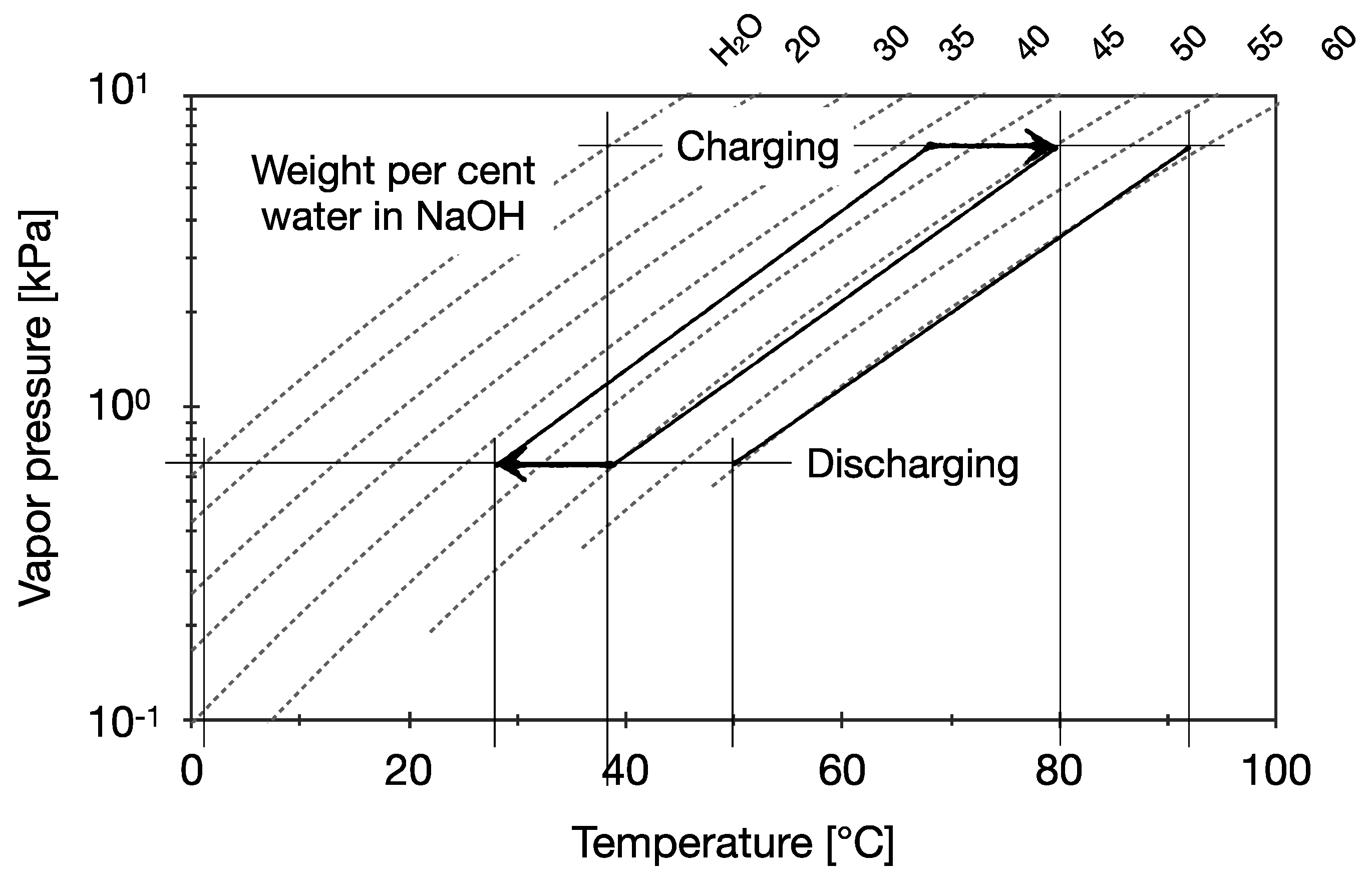
Energies | Free Full-Text | Static Temperature Guideline for Comparative Testing of Sorption Heat Storage Systems for Building Application